|
7/21/2023 0 Comments Workdone in thermodynamics 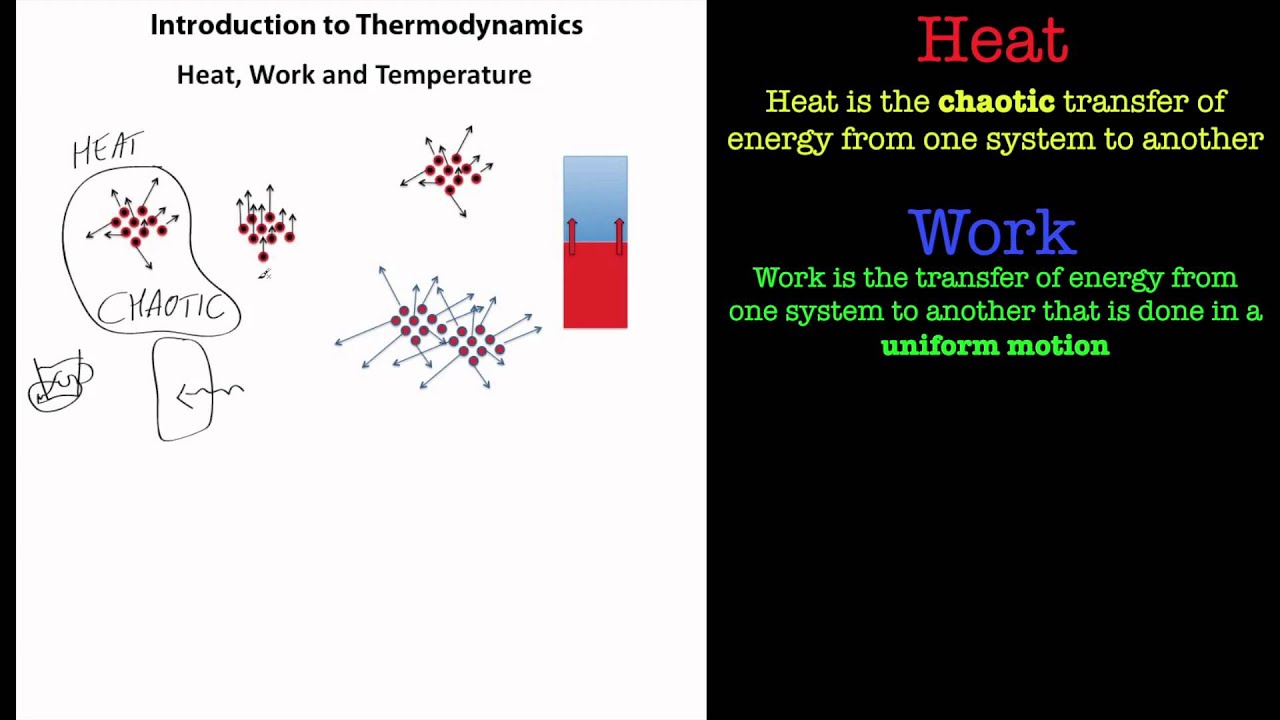
Real gases differ from these in certain ways. Inform students that the ideal gas law is only strictly true for gases that are ideal. This is why railroad tracks and bridges have expansion joints that allow them to freely expand and contract with temperature changes. Gases are especially affected by thermal expansion, although liquids expand to a lesser extent with similar increases in temperature, and even solids have minor expansions at higher temperatures. What is the underlying cause of thermal expansion? An increase in temperature means that there’s an increase in the kinetic energy of the individual atoms. This last point describes thermal expansion-the change in size or volume of a given mass with temperature. When pressure is constant, volume is directly proportional to temperature.When temperature is constant, pressure is inversely proportional to volume.When volume is constant, pressure is directly proportional to temperature.Instead, it is important for us to notice from the equation that the following are true for a given mass of gas: The constant k is called the Boltzmann constant and has the value k = 1.38 × 10 −23 J/K, k = 1.38 × 10 −23 J/K, For the purposes of this chapter, we will not go into calculations using the ideal gas law. Where P is the pressure of a gas, V is the volume it occupies, N is the number of particles (atoms or molecules) in the gas, and T is its absolute temperature.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
